Systems Microbiology of Intracellular Pathogens

Our Research
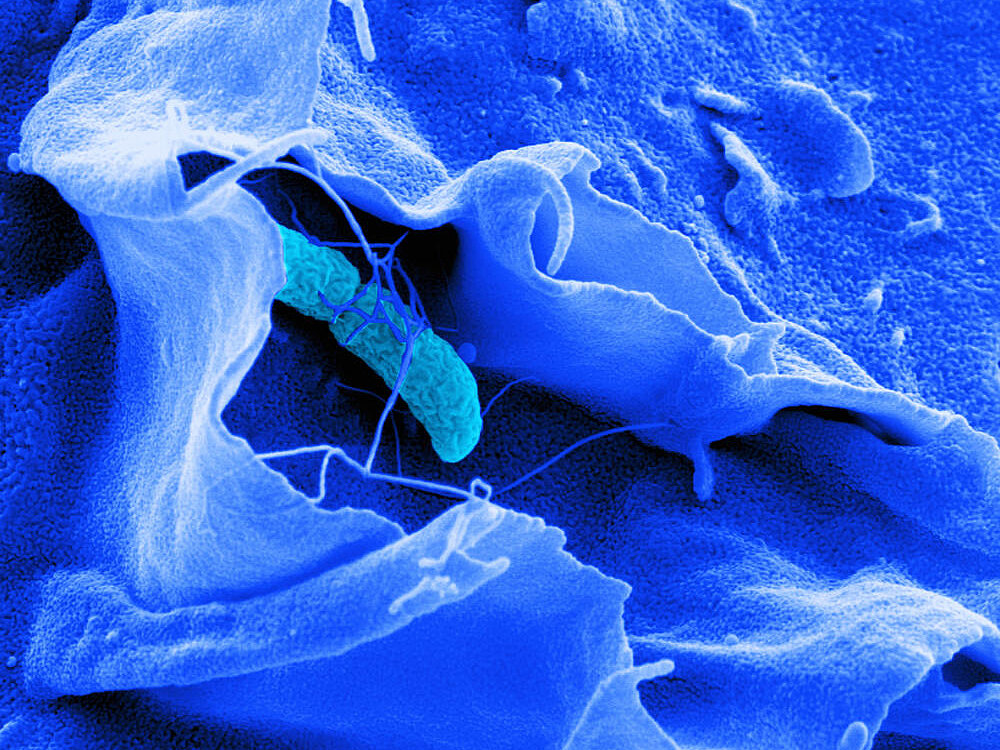
Antibiotic-resistant intracellular pathogens pose a critical threat: they hide inside our cells, evading both immune defenses and antibiotics. Understanding how these bacteria adapt and survive requires capturing the full picture of host-pathogen interactions at single-cell resolution in physiologically-relevant infection models—something current technologies cannot achieve.
Camilla Ciolli Mattioli’s group develops innovative platforms that simultaneously capture host and pathogen gene expression at single-cell resolution during infection. The researchers complement this with spatial transcriptomics to map interactions within native tissue architectures and CRISPR-based molecular recording systems to trace bacterial transcriptional histories.
By integrating spatial, temporal, and phenotypic dimensions—using image-enabled cell sorting to isolate specific infection outcomes—the group identifies molecular switches that determine whether bacteria persist, replicate, or face elimination. Its goal is to discover vulnerabilities in bacterial survival strategies and reveal therapeutic targets that redirect infections toward pathogen clearance, advancing the fight against antimicrobial resistance.
Our Research
Antibiotic-resistant intracellular pathogens pose a critical threat: they hide inside our cells, evading both immune defenses and antibiotics. Understanding how these bacteria adapt and survive requires capturing the full picture of host-pathogen interactions at single-cell resolution in physiologically-relevant infection models—something current technologies cannot achieve.
Camilla Ciolli Mattioli’s group develops innovative platforms that simultaneously capture host and pathogen gene expression at single-cell resolution during infection. The researchers complement this with spatial transcriptomics to map interactions within native tissue architectures and CRISPR-based molecular recording systems to trace bacterial transcriptional histories.
By integrating spatial, temporal, and phenotypic dimensions—using image-enabled cell sorting to isolate specific infection outcomes—the group identifies molecular switches that determine whether bacteria persist, replicate, or face elimination. Its goal is to discover vulnerabilities in bacterial survival strategies and reveal therapeutic targets that redirect infections toward pathogen clearance, advancing the fight against antimicrobial resistance.
Jun Prof Camilla Ciolli Mattioli
What determines whether intracellular bacteria persist, replicate, or are cleared within the host remains a fundamental question in infection biology.
Camilla Ciolli Mattioli studied biotechnology at the University of Florence (Italy) and received her PhD from the Humboldt University of Berlin (Germany) in 2019. During her doctoral research at the Berlin Institute for Medical Systems Biology of the Max Delbrück Center, she focused on the mechanisms driving RNA localization and local translation in eukaryotic cells. She then won a Marie-Curie fellowship and joined the lab of Roi Avraham at the Weizmann Institute of Science (Rehovot, Israel) as a postdoctoral fellow, where she investigated how bacterial phenotypic heterogeneity affects infection outcomes. She has been a research group leader at the Helmholtz Institute for RNA-based Infection Research (HIRI) and a junior professor at the University of Würzburg since February 2026.
Selected Publications
Ciolli Mattioli C (2026) Hedging their bets: how bacterial pathogens diversify to survive infection. Current Opinion in Microbiology 91:102729 DOI: 10.1016/j.mib.2026.102729
Persky Z, Ciolli Mattioli C, Garcia KZ, Azran O, Avraham R, Dar D (2026) Single-cell transcriptome imaging reveals conserved and virulence-linked phenotypic states in bacteria bioRxiv DOI: 10.64898/2026.02.01.703127
Ciolli Mattioli C, Eisner K, Rosenbaum A, Wang M, Rivalta A, Amir A, Golding I, Avraham R (2023) Physiological stress drives the emergence of a Salmonella subpopulation through ribosomal RNA regulation. Current Biology 33(22):4880-4892.e14 DOI: 10.1016/j.cub.2023.09.064
Heyman O, Yehezkel D, Ciolli Mattioli C, Blumberger N, Rosenberg G, Solomon A, Hoffman D, Bossel Ben-Moshe N, Avraham R (2023) Paired single-cell host profiling with multiplextagged bacterial mutants reveals intracellular virulence-immune networks. PNAS 120(28):e2218812120 DOI: 10.1073/pnas.2218812120
Rosenberg G, Yehezkel D, Hoffman D, Ciolli Mattioli C, Fremder M, Ben-Arosh H, Vainman L, Nissani N, Hen-Avivi S, Brenner S, Itkin M, Malitsky S, Ohana E, Ben-Moshe NB, Avraham R (2021) Host succinate is an activation signal for Salmonella virulence during intracellular infection. Science 371(6527):400-405 DOI: 10.1126/science.aba8026
A complete list of publications can be found here.