Personalised Immunotherapy

Our Research: Methods & vaccines 2.0
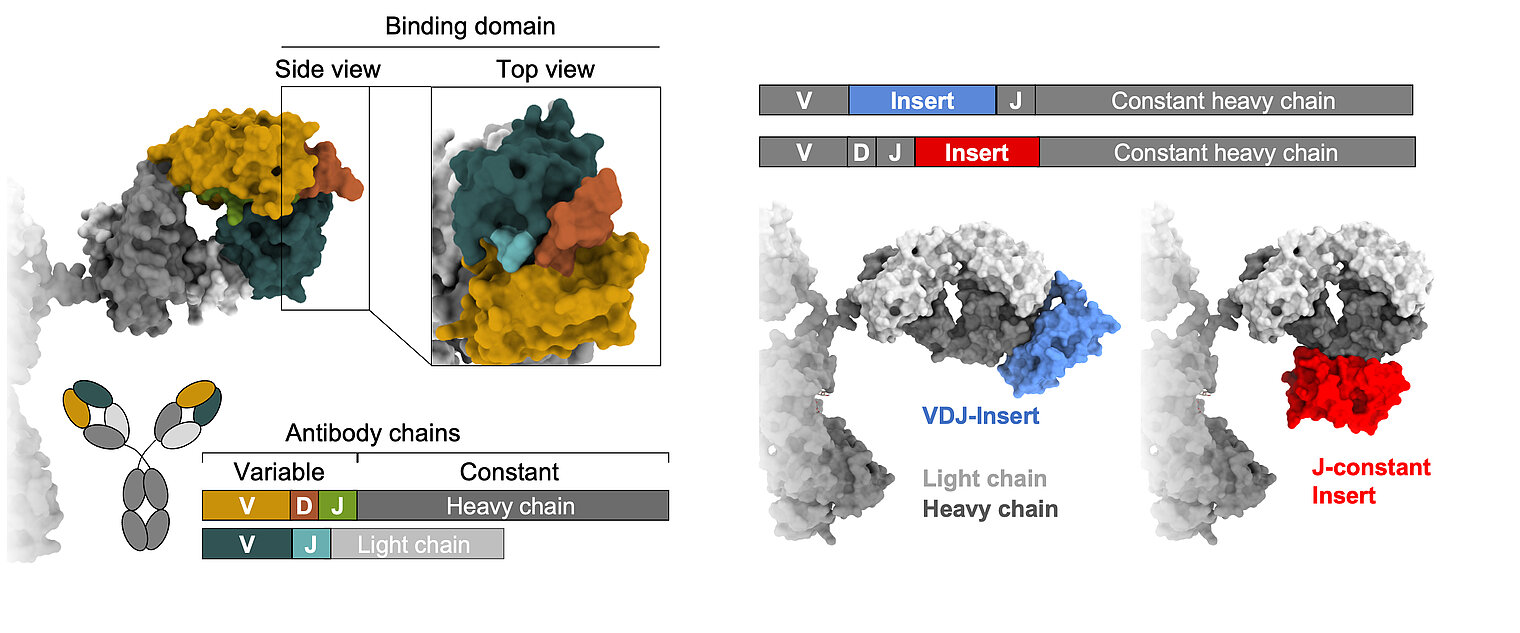
Learning from nature, we strive to gain a comprehensive understanding of B cell diversification and function in humans. In addition to classical recombination events, we study a rare but sometimes particularly useful diversification process: the acquisition of large DNA inserts in the heavy chain genes that equip antibodies with extra domains. Our research is based on three main pillars:
Our first research goal is to develop novel tools to study the repertoire of antibodies and the underlying recombination events. One of our methods holds promise for the personalized assessment of genomic scarring. Prospectively, a biomarker for DNA repair may be used for the prediction and prognosis of diseases related to DNA-repair malfunction, such as defects of the immune system.
Our second goal is to develop personalized cell-based vaccines. After identifying the blind spots in our B cell repertoire, B cells “by design” may extend the natural repertoire through artificial immunity to endow the body with antibodies of superior reactivity.
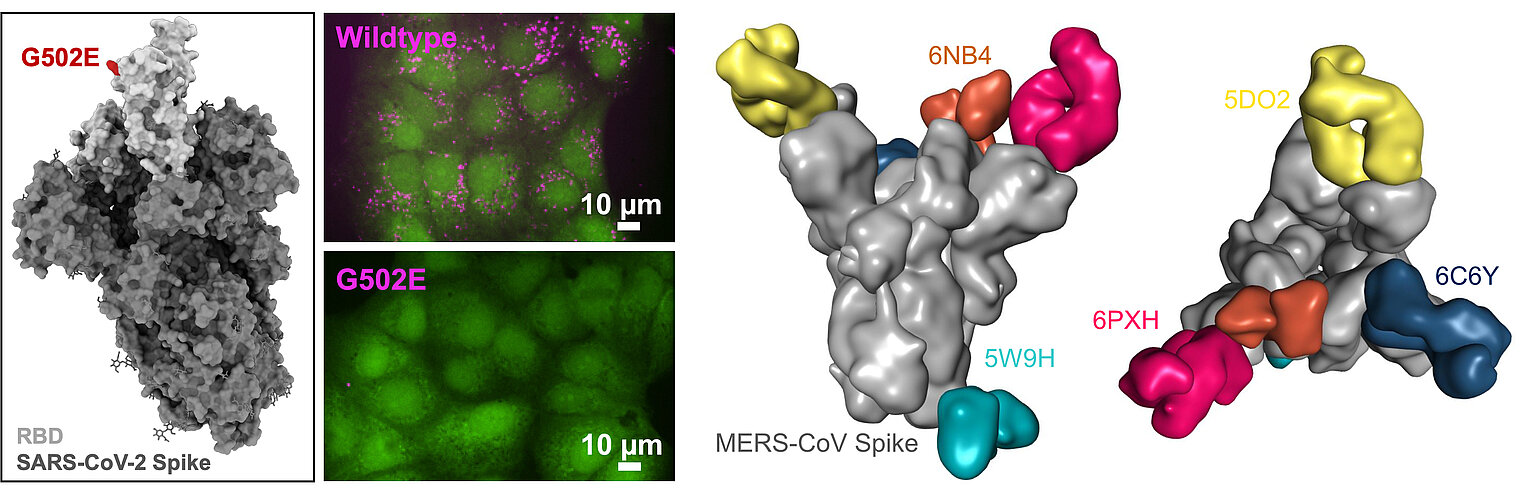
The third branch of our research arose during the SARS-CoV-2 pandemic. We aim to improve the safety and efficacy of vaccines by preventing their interaction with body-own structures via so-called Body-Inert, but B cell Activating vaccines (BIBAX). In the future, we aim to explore how interactions of vaccines with body receptors hamper immune responses to pathogens.
Our Research: Methods & vaccines 2.0
Learning from nature, we strive to gain a comprehensive understanding of B cell diversification and function in humans. In addition to classical recombination events, we study a rare but sometimes particularly useful diversification process: the acquisition of large DNA inserts in the heavy chain genes that equip antibodies with extra domains. Our research is based on three main pillars:
Our first research goal is to develop novel tools to study the repertoire of antibodies and the underlying recombination events. One of our methods holds promise for the personalized assessment of genomic scarring. Prospectively, a biomarker for DNA repair may be used for the prediction and prognosis of diseases related to DNA-repair malfunction, such as defects of the immune system.
Our second goal is to develop personalized cell-based vaccines. After identifying the blind spots in our B cell repertoire, B cells “by design” may extend the natural repertoire through artificial immunity to endow the body with antibodies of superior reactivity.
The third branch of our research arose during the SARS-CoV-2 pandemic. We aim to improve the safety and efficacy of vaccines by preventing their interaction with body-own structures via so-called Body-Inert, but B cell Activating vaccines (BIBAX). In the future, we aim to explore how interactions of vaccines with body receptors hamper immune responses to pathogens.
“Our mission is to understand the universe of the antibody repertoire. Our vision is to transform this knowledge into personalized solutions for infectious diseases.”

Kathrin de la Rosa (born Kathrin Pieper in 1984) became an immunologist after completing her doctoral studies in 2013 at the University Medical Center Freiburg on B cell disorders in immunodeficient patients. As a postdoctoral fellow, she decided to study monoclonal antibodies in infectious diseases. To this end, she joined the research group of Antonio Lanzavecchia at the Institute for Research in Biomedicine in Switzerland. In 2018, she started her own research group at the Max Delbrück Center in the Helmholtz Association (MDC) in Berlin after being awarding the Emmy Noether Fellowship of the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG). In 2020, she was awarded the European Research Council (ERC) starting grant and was selected to obtain the Johanna Quandt Fellowship of the Stiftung Charité. She became a W2 Professor of “Immune mechanisms in translation” at the Berlin Institute of Health@Charité in 2021 and, since 2024, is a W3 Professor of Personalized Immunotherapy (W3) at the Hannover Medical School (MHH) in a joint appointment with the Helmholtz Centre for Infection Research (HZI). She is running her research group at the Centre for Individualised Infection Medicine (CiiM).
Team











Guests and former members
Former doctoral students (including guests; if they obtained their doctorate abroad or at other German research institutions, the universities/institutes where they worked are listed)
Former postdoctoral researchers and visiting doctoral candidates (including researchers who received funding from AvH, DAAD, and other scholarships)
- Irina Yakuteno
- Carlotta Caramel
- Mateja Zvipelj
Former (visiting) students
- Miriam Herfort
- Dunja Vukcevic
Former technical employees
- Franziska Flohr
Selected Publications
- Clara Vázquez García, Benedikt Obermayer, Baerbel Keller, Mikhail Lebedin, Christoph Ratswohl , Hassan Abolhassani, Antonia Busse, Michela Di Virgilio, Stephan Mathas, Dorothee Speiser, Dieter Beule, Qiang Pan-Hammarström, Klaus Warnatz, Kathrin de la Rosa. Recombination junctions from antibody isotype switching classify immune and DNA repair dysfunction. Nature Communications DOI: 10.1038/s41467-025-67206-5 PMID: 41419734 Open Access
- Mikhail Lebedin, Nikolai Petrovsky, Kairat Tabynov, Kaissar Tabynov, Yuri Lebedin. SARS-CoV-2 neutralization and protection of hamsters via nasal administration of a humanized neutralizing antibody. Antiviral Research DOI: 10.1016/j.antiviral.2025.106235 PMID: 40645296 Open Access
- Likun Du, Valentyn Oksenych, Hui Wan, Xiaofei Ye, Junchao Dong, Adam Yongxin Ye, Hassan Abolhassani, Stelios Vlachiotis, Xuefei Zhang, Kathrin de la Rosa, Lennart Hammarström, Mirjam van der Burg, Frederick W Alt, Qiang Pan-Hammarström. Orientation Regulation of Class-switch Recombination in Human B Cells. The Journal of Immunology DOI: 10.4049/jimmunol.2300842 PMID: 39248600
- Mikhail Lebedin, Kathrin de la Rosa. Diversification of Antibodies: From V(D)J Recombination to Somatic Exon Shuffling. Annual Review Cell and Developmental Biology DOI: 10.1146/annurev-cellbio-112122-030835 PMID: 39356809 Open Access
- Lebedin, M., C. Ratswohl, A. Garg, M. Schips, C. V. García, L. Spatt, C. Thibeault, B. Obermayer, J. Weiner, I. M. Velásquez, C. Gerhard, P. Stubbemann, L.-G. Hanitsch, T. Pischon, M. Witzenrath, L. E. Sander, F. Kurth, M. Meyer-Hermann*, and K. de la Rosa*. 2024. Soluble ACE2 correlates with severe COVID-19 and can impair antibody responses. iScience 27: 109330. DOI: 10.1016/j.isci.2024.109330. PMID: 38496296. Open Access
Publications
A complete list of publications can be found in the ORCID profile of Kathrin de la Rosa.
Major Grants and Consortia
CoViPa – Helmholtz Network Fund (2021-2025)
CoViPa is a joint research initiative of 7 Helmholtz institutes and 3 university partners. Together with the German Aerospace Center (DLR, Wessling) and the German Cancer Research Center (DKFZ, Heidelberg) we aim develop targeted immunomodulatory strategies by means of AI-based robotic platforms.
Johanna Quandt Fellowship of the Stiftung Charité (2021-2026)
The goal of this program is to develop and study innovative approaches for two challenges of translational immunology: cellular vaccines for improved humoral immunity and the use antibodies to modulate T cell responses.
Starting grant of the European research council (ERC) (2020-2025)
This research project aims to develop novel strategies for B cell engineering exploiting natural DNA-breaks to generate antibodies that surpass common reactivity profiles.
Emmy Noether Project of the german research foundation (2018-2024)
The project aims to study a new layer of antibody diversity that is generated by integration of large DNA inserts in the antibody heavy chain locus.