RSV causes respiratory infections that can be particularly severe in young children and older people. According to the World Health Organisation (WHO), around 3.6 million children under the age of five are treated in hospital for RSV infections every year, and 100,000 of them die as a result of the infection. Currently, the only treatment available is supportive care, such as oxygen therapy. The consortium therefore aims to develop new treatment options as part of the “OPERA” (optimising pan-entry RSV antivirals) project.
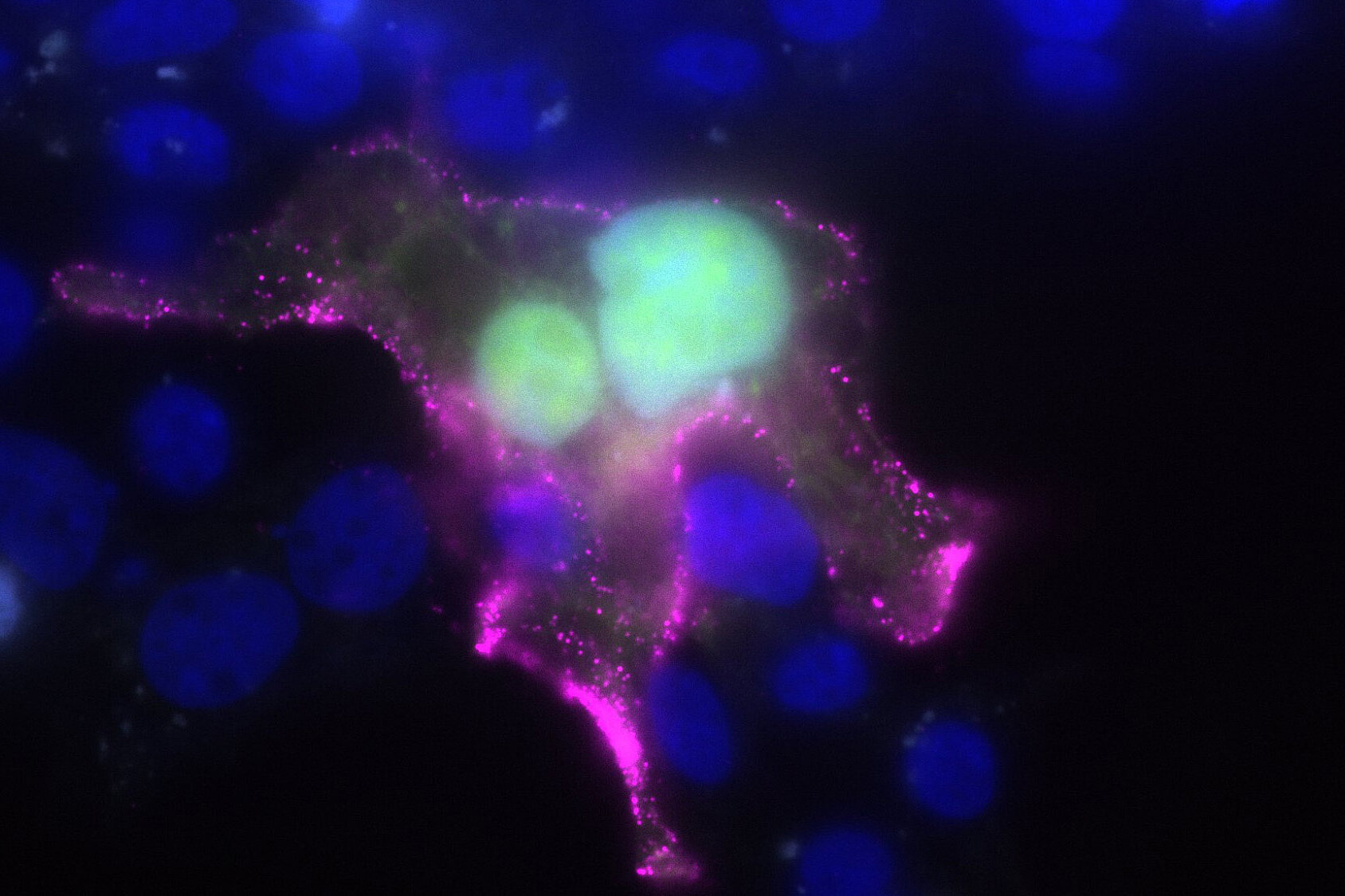
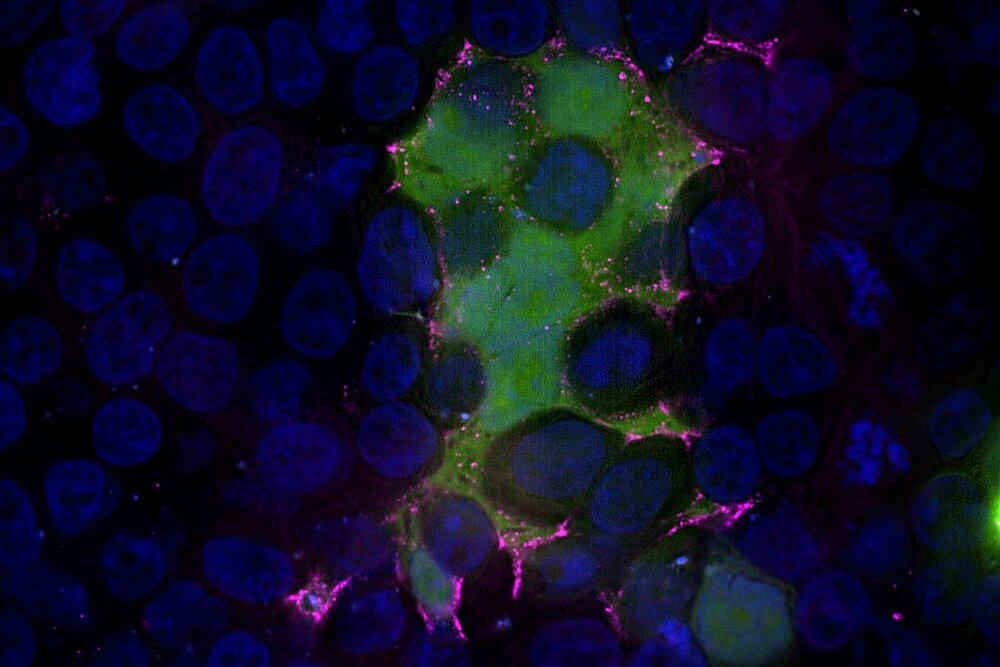
”In order to enter human cells, the virus must fuse with the membrane,” says Prof. Pietschmann. He is director of the Institute for Experimental Virology at TWINCORE in Hanover, head of the department of the same name at HZI and heads the research project. The fusion protein, or F protein for short, plays an essential role in this process. In previous work, the research team has identified inhibitors that can very effectively inhibit this step of infection at the cell surface. However, they are not very effective against alternative fusion inside the cell, which likely occurs in an environment with lower pH. “That's why we want to optimize the fusion inhibitors so that they are active in a wider pH range,” says Prof. Anna Hirsch, head of the department “Drug Design and Optimisation” at HIPS. “This would significantly improve the effectiveness of the inhibitors.”
In addition, a formulation suitable for inhalation is to be developed. “This will allow the active ingredient to reach the lungs quickly and in a targeted manner,” says Prof. Martin Empting, who heads the research group “Antiviral and Antivirulence Drugs” at HIPS. The active ingredient will first be comprehensively tested in human primary cells. Additionally analyzing absorption, distribution, metabolism, excretion and toxicity (ADMET) allows to understand and optimize pharmacokinetics and pharmacodynamics (PK/PD) of the substance. Its efficacy will then be demonstrated in animal models. “This is the milestone for the transition to industrial development,” says Dr Katharina Rox, head of the research group “Pharmacokinetics and Pharmacodynamics” at the HZI. Once the defined milestones have been reached, the industrial partner ENYO Pharma SA will take over the preclinical development up to the first human studies.
In addition to Prof. Thomas Pietschmann's team, which is conducting the virological profiling, various other groups are involved in this project. The teams led by Prof. Anna Hirsch and Prof. Martin Empting at the Helmholtz Institute for Pharmaceutical Research Saarland (HIPS) are contributing their expertise in the fields of medicinal chemistry and computer-aided drug optimization. Prof. Thomas Krey's laboratory at the University of Lübeck is working on the structural biology aspects of the F protein. Dr Katharina Rox is conducting the pharmacological studies at the HZI in Braunschweig. Prof. Gesine Hansen, Medical Director of the Clinic for Pediatric Pneumology, Allergology and Neonatology at Hannover Medical School, is responsible in particular for demonstrating the in vivo proof of concept in a lung infection model. The biotechnology company ENYO Pharma SA from Lyon, France, is responsible for the subsequent industrial development.
The Translational Project Management Office (TPMO) of the German Center for Infection Research (DZIF) supports the consortium and accompanies the project with professional, translational project management by Dr Marina Steindorff.