Malaria – tiny bite, major effect
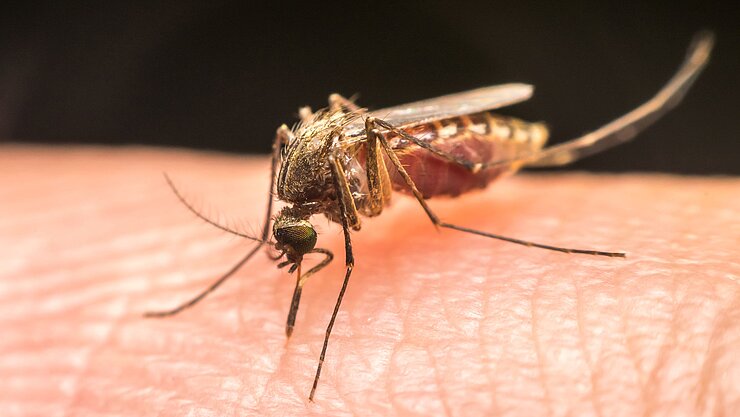
Along with tuberculosis and AIDS, malaria is one of the “big three”, the three infectious diseases that are globally responsible for most deaths. According to World Health Organization estimates, some 228 million cases of malaria were reported in 2018 alone. Children are at an especially high risk as their immune system is not yet familiar with the disease. In Africa, every second minute, a child dies from malaria.
The risk of infection is highest at night as this is the prime time for the Anopheles mosquito to bite and, simultaneously, transmit Plasmodium, the malaria pathogen. An infected female mosquito namely not only sucks blood - it also injects sporozoites, which represent one of the infective forms of the single-celled parasite.
One parasite, many life forms
In humans and the Anopheles mosquito, Plasmodium goes through different developmental stages. A mosquito bite transmits the sporozoites into the human circulation where the parasite initially develops and proliferates inside liver cells and, thereafter, inside red blood cells, also known as erythrocytes, which supply vital oxygen to our cells. Inside erythrocytes, Plasmodium matures to its sexually reproductive gametocyte stage. If, during a blood meal, the Anopheles mosquito feeds on an infected person's blood, red blood cells containing gametocytes end up in the midgut of the mosquito, the site of sexual reproduction. The life cycle completes a full circle when the pathogen migrates to the salivary glands of the mosquito, ready to infect its next victim. This way, the mosquito has effectively become a "vector", transmitting the pathogen - and with it the disease - from person to person.
Special climatic conditions are required for the proliferation of the Anopheles mosquito and for Plasmodium to go through the different stages of its life cycle. Ideal conditions exist in tropical and subtropical regions. Over 40 % of world’s population lives in these malaria endemic areas.
Malaria - the “swamp fever”
During the parasite life cycle, infected erythrocytes burst on a regular basis, often simultaneously, allowing the released parasites to infect new red blood cells. Along with the single-celled parasites, toxic metabolites end up in the bloodstream, leading to periodic fevers, nausea, and headaches – the typical symptoms of malaria that can be traced back to the immune response. This inflammatory response is significantly abated in patients who are, on one set of their chromosomes, carriers of the gene for a disease called sickle cell anemia. In these individuals, part of their oxygen-carrying proteins are structurally defective, and some erythrocytes assume a sickled shape, hence the name of the disease. As the underlying mutation protects carriers from malaria, sickle cell anemia is highly prevalent in areas where malaria is endemic.
Different Plasmodium species cause malaria in humans
There are four known species of Plasmodium that can cause malaria in humans: Plasmodium falciparum, Plasmodium vivax, Plasmodium malariae, and Plasmodium ovale. One other species, Plasmodium knowlesi, primarily infects monkeys but can also affect humans. Plasmodium falciparum, the pathogen that causes Malaria tropica, is considered especially dangerous as it leads to a range of complications. Left untreated, this form is often fatal.
To date, no approved vaccine exists, although a number of candidate vaccines are currently undergoing testing in clinical trials. Consequently, the best prevention is to avoid getting bitten by the mosquito using protection, such as mosquito nets and insecticide sprays. Since resistance to standard therapeutics is commonly observed, the need for research on novel drug candidates is urgent.
One of the things HZI scientists are interested in is how the parasite moves and enters its host cells. With the help of biophysical, biochemical, and structural biology methods, our scientists are studying the parasite’s cytoskeleton, which forms a dynamic internal scaffold that is crucial in the process of motility and invasion. One of their methods involves bombarding highly purified and crystallized cytoskeletal components with X-rays that are produced when charged particles are radially accelerated in a strong magnetic field. Such particle accelerator-based experiments reveal the atomic structures of the proteins that are involved and may provide the basis for development of new malaria drugs.
(bma)